Ethics Application Process
To apply for research ethics approval, you will need to submit an application via the NSW Research Ethics and Governance Information System (REGIS). Quick Reference Guides and walk-through videos can be found on the REGIS How-To page under Applicants - ETHICS: APPLICATION (Initial submission - pre-approval).
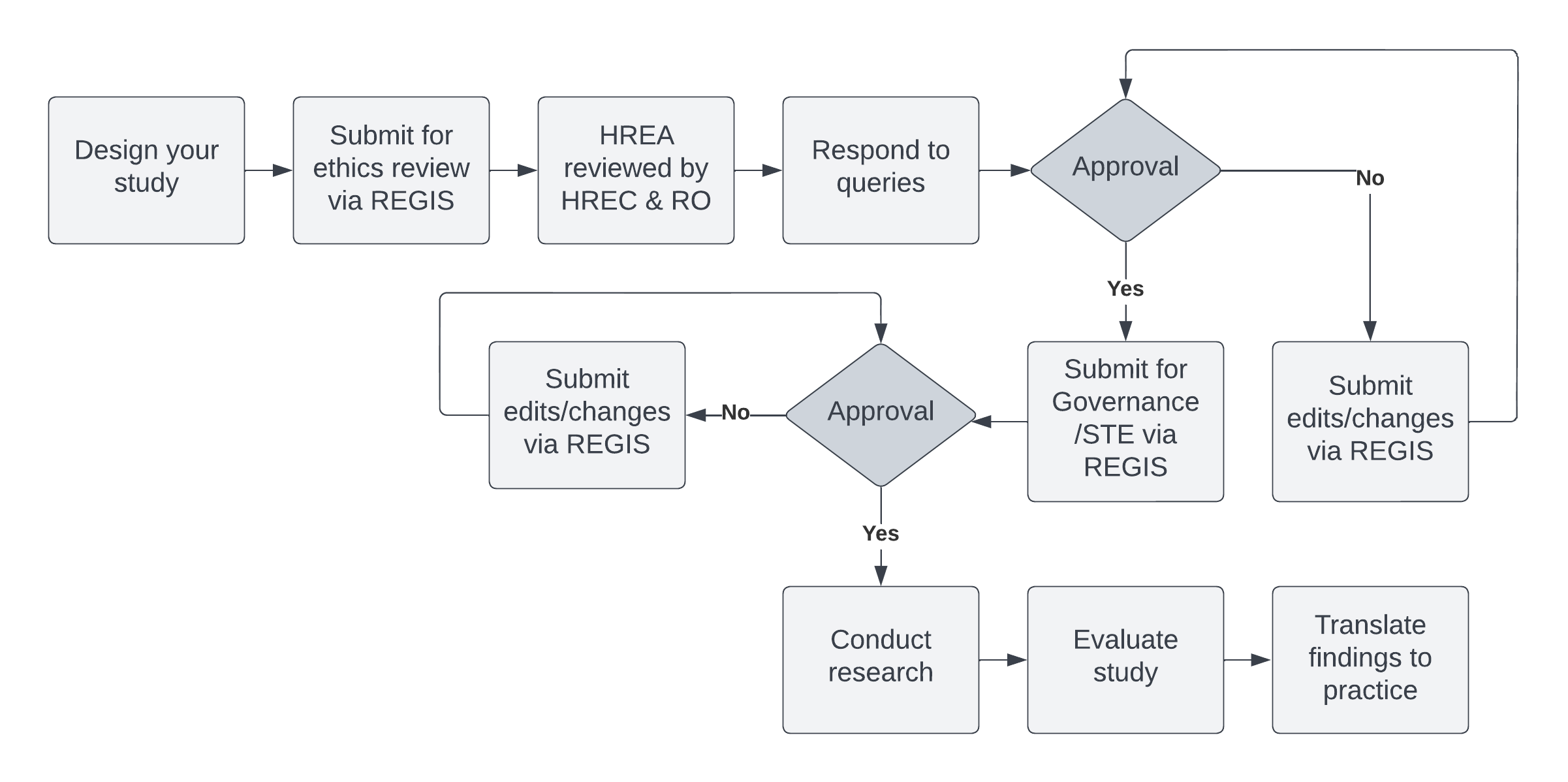
All applications to the HREC must comply with the Ethics Eligibility Checklist and must be uploaded with your application. All applications must also include an Human Research Ethics Application (HREA) (completed within REGIS), a Study Protocol, a Method of Payment Form and any other study documents as necessary for your project i.e. Participant Information Sheet and Consent Forms, Data Collection Sheet, study questionnaires, advertising material, Victorian Specific Module, Radiation Safety Report.
All documents are to be uploaded in REGIS. Please refer to the Quick Reference Guides (Project Registration, HREA/Ethics Application - Completing and Submitting the Application). All study documents must have a version number and date in the footer.
In the HREA, you will be required to nominate a review pathway. This is determined by a project's level of risk (i.e. Low Risk or Greater than Low Risk). If you are uncertain of which pathway your research falls under, please see the Low and Negligible Risk page or contact the Research Office. Low Risk Applications are reviewed via the Low Risk Ethics Committee.
Please ensure information provided on the documentation is consistent. For example, information provided in the Protocol must match what is included in the HREA. Inconsistent information across the application will result in the application being returned for amending.
Please contact the Research Office if you have any questions about your research project or application in REGIS. For any technical support, call the REGIS Help Desk on 1300 073 447.
What is an Human Research Ethics Application (HREA)?
A Human Research Ethics Application (HREA) is a web-based form developed by the National Health and Medical Research Council (NHMRC) to assist researchers consider and address the ethical principles set out by the National Statement on Ethical Conduct in Human Research (2007). In NSW, HREA are completed by the REGIS platform. Quick Reference Guides are available to assist researchers in preparing their HREA and submitting through the REGIS.
Please note, when preparing an HREA it is important you provide all relevant details and documentation. This includes ensuring details are accurate and consistent across ALL documentation you have provided. For example, if you propose to conduct a study over multiple sites, you will need to list each site on the project registration, protocol and HREA. Similarly, you will need to ensure all the names and roles of the research team are consistent across documentation too.
SESLHD Human Research Ethics Committee (HREC)
The SESLHD Human Research Ethics Committee (HREC) is an accredited lead in administering independent review of HREAs. The committee functions to promote ethical research across the district and protect the rights, dignity and safety of human participants. This is achieved through providing ethical oversight, monitoring and advice to researchers, as well as prescribing principles and procedures to safely govern research projects.
It is important to keep abreast the HREC meeting and submission dates to avoid delay in project review and approval. In particular, when the HREC has queries about an application, it may delegate correspondence to an Executive Committee or Executive Officer. If additional information needs to be reviewed by the Executive Committee, please be aware you must submit changes/amendments by the stipulated submission dates.
Responses to Committee queries
When responding to queries:
- Please submit your reply via REGIS. Quick Reference Guides and walk-through videos are available under Applicants - ETHICS: APPLICATION (Initial submission - pre-approval)
- Please include a cover letter which addresses (point-wise) the questions raised by the committee and your response. Both the questions and your response should be included in the cover letter.
- Please amend documents in accordance with the requests and provide documents with tracked changes in addition to a clean copy.
- Please include and update the version number, date and page number in the footer of all documents i.e. Version 3 01/01/2022, page 2 of 5
If you would like to discuss your application and/or HREC queries, you can contact the SESLHD Research Office.
Does my Project require Ethical Review?
NSW Health adopts the distinctions between projects requiring ethical review and those that do not as set out by the NSW Health Guideline: GL2007_020 - Human Research Ethics Committees - Quality Improvement & Ethical Review: A Practice Guide for NSW.
Researchers conducting human research are required to submit their projects to a Human Research Ethics Committee as per NSW Health Policy.
Quality Assurance (QA)
NSW Health encourages the widespread implementation of Quality Assurance (QA) programs as an essential part of a learning healthcare organisation. QA should be conducted as part of conventional clinical service delivery or 'business-as-usual'. In most routine situations, QA projects should not require ethical review.
QA practitioners should complete a Research Office QA Form which will assist in assessing the characteristics of their project against the NSW Health Guidelines to determine if any of the ethical risks described within are present in their project.
If ethical risks are identified, review by an ethics review body should be sought. Please follow the instructions in the QA form regarding submission to the Research Office. If ethical risks are not identified, QA practitioners are free to commence their projects, after adhering to any institutional requirements. NSW Health policy outlines ethics review and approval is not required merely for the mechanism of obtaining publication in a journal [see Q16 in GL 2007_020].
For more information on QA initiatives, please visit the OHMR website.
Guidance for Clinical Case Report Submission
In keeping with the requirements of some journals, the SESLHD HREC supports a process by which case report proposals are reviewed by an ethical sub-committee. The committee requests that the below form is completed and submitted to SESLHD-RSO@health.nsw.gov.au (subject title: Case Report Submission). A blank copy of the consent form to be used to gain consent from the clinical case must also be submitted for review. Some journals require the use of a specific consent form. A template has also been provided below:
The committee will review your case report for ethical risks and if none are identified will provide you with correspondence confirming that full ethical review is not required. This correspondence may be provided to the editors of Journals requesting clearance from an Ethical Review Committee but is not a statement of ethical approval.